The main difference between BenevolentAI, Atomwise and Insilico is that BenevolentAI concentrates its AI leverage on upstream target and indication discovery using knowledge‑graph‑driven biology, Atomwise specialises in structure‑based virtual screening for small‑molecule hits, and Insilico spans an end‑to‑end, generative AI pipeline that connects target discovery, de novo design and experimental validation through an autonomous lab and a growing clinical‑stage portfolio
All data and examples in this comparison are drawn from HealthyData.Science’s independently researched directory profiles for BenevolentAI, Atomwise and Insilico. [1]
Table 1: At a glance – How the three drug discovery AI platforms create value
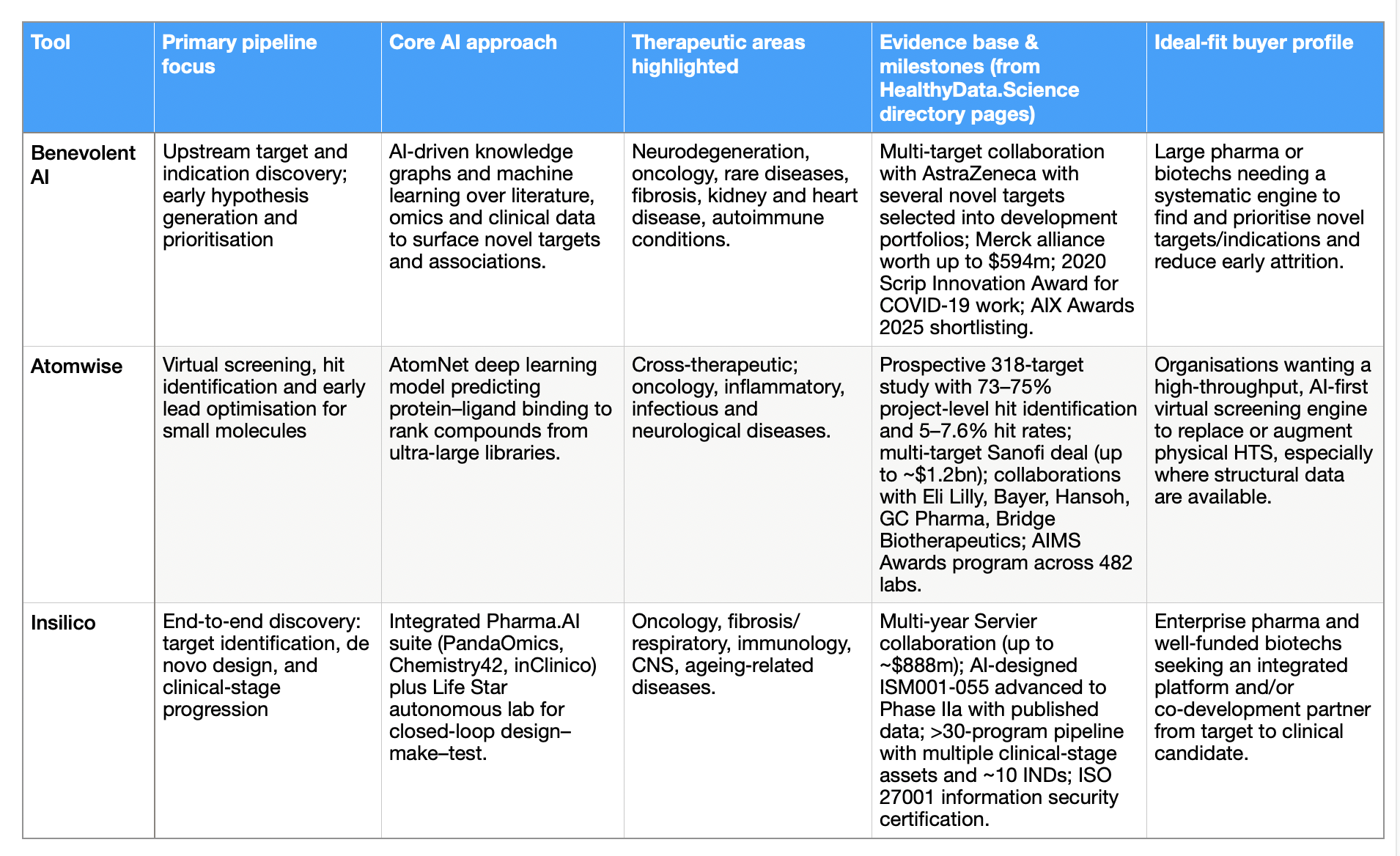
Table 2: What buyers should ask – and how each AI tool answers
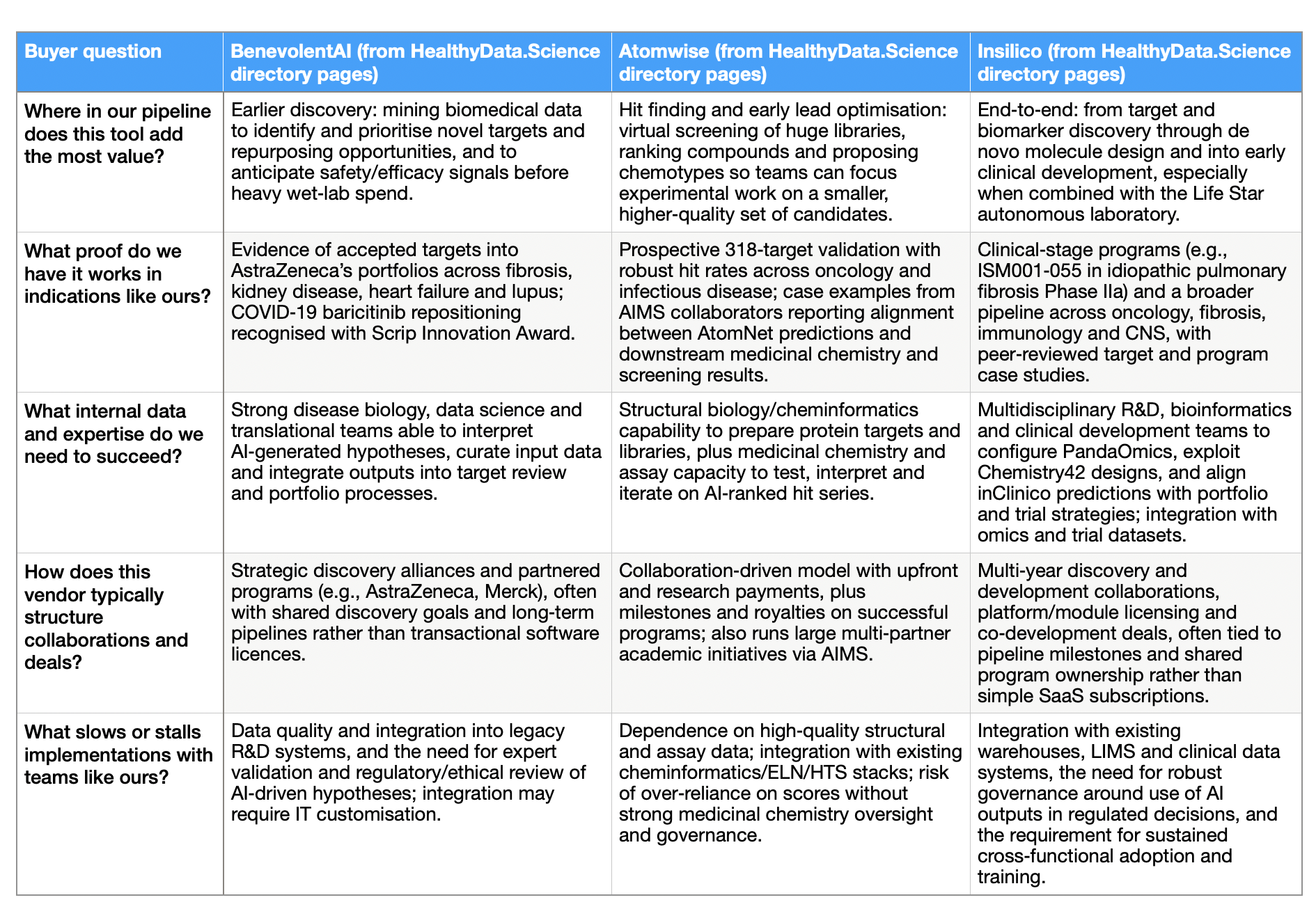
Table 3: Risks and deal‑stallers across the three platforms
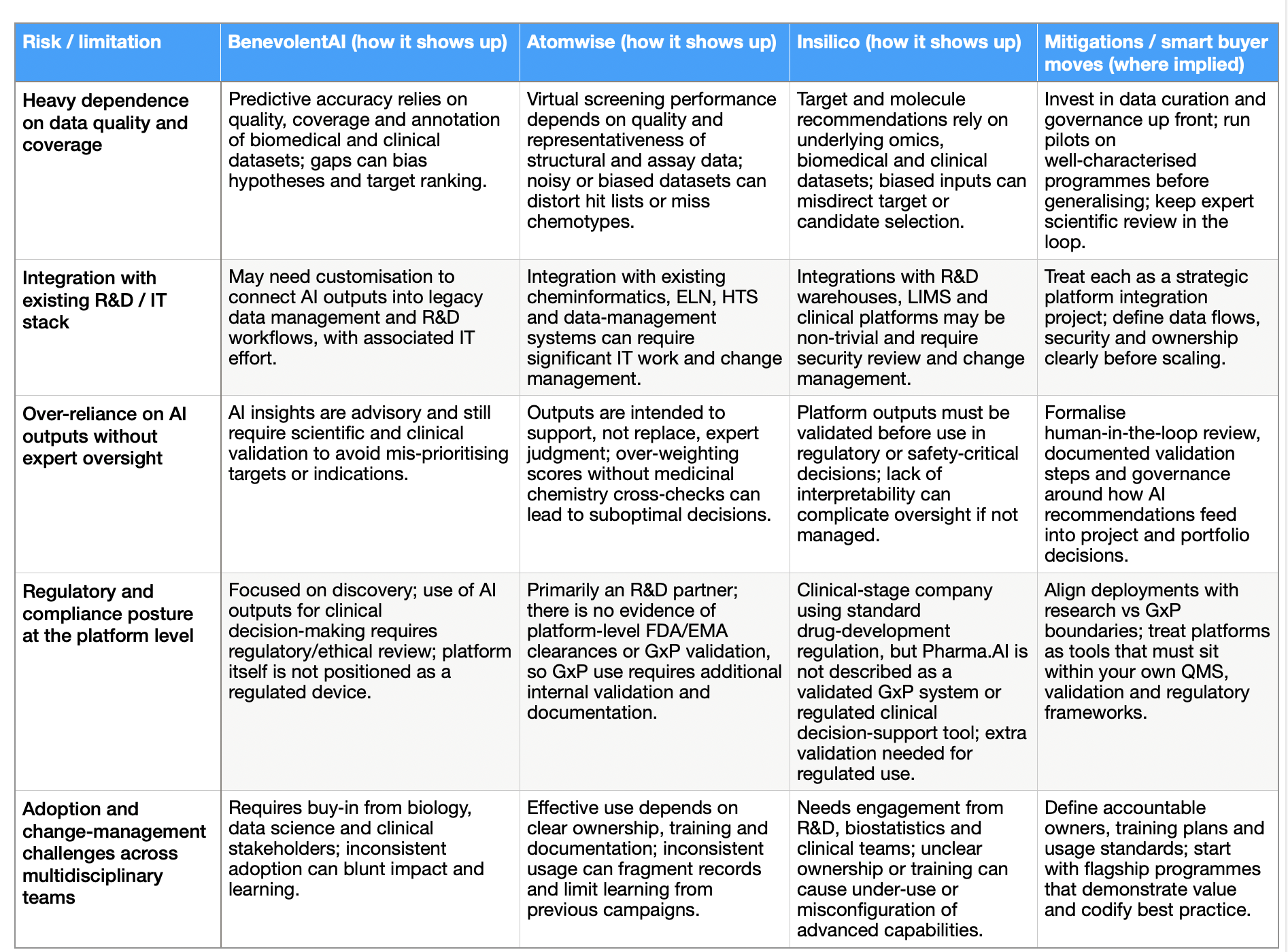
BenevolentAI: upstream biology and portfolio‑grade hypotheses
BenevolentAI is positioned as an AI‑driven discovery engine that mines scientific literature, clinical trials and real‑world data to uncover novel disease biology and therapeutic hypotheses that traditional methods may miss. Its knowledge‑graph and machine‑learning stack is designed to generate and prioritise targets, indications and repurposing opportunities, with use cases ranging from idiopathic pulmonary fibrosis and chronic kidney disease to heart failure, lupus, oncology and rare diseases.
Figure 1: BenevolentAI: upstream biology and portfolio-grade hypotheses [2]
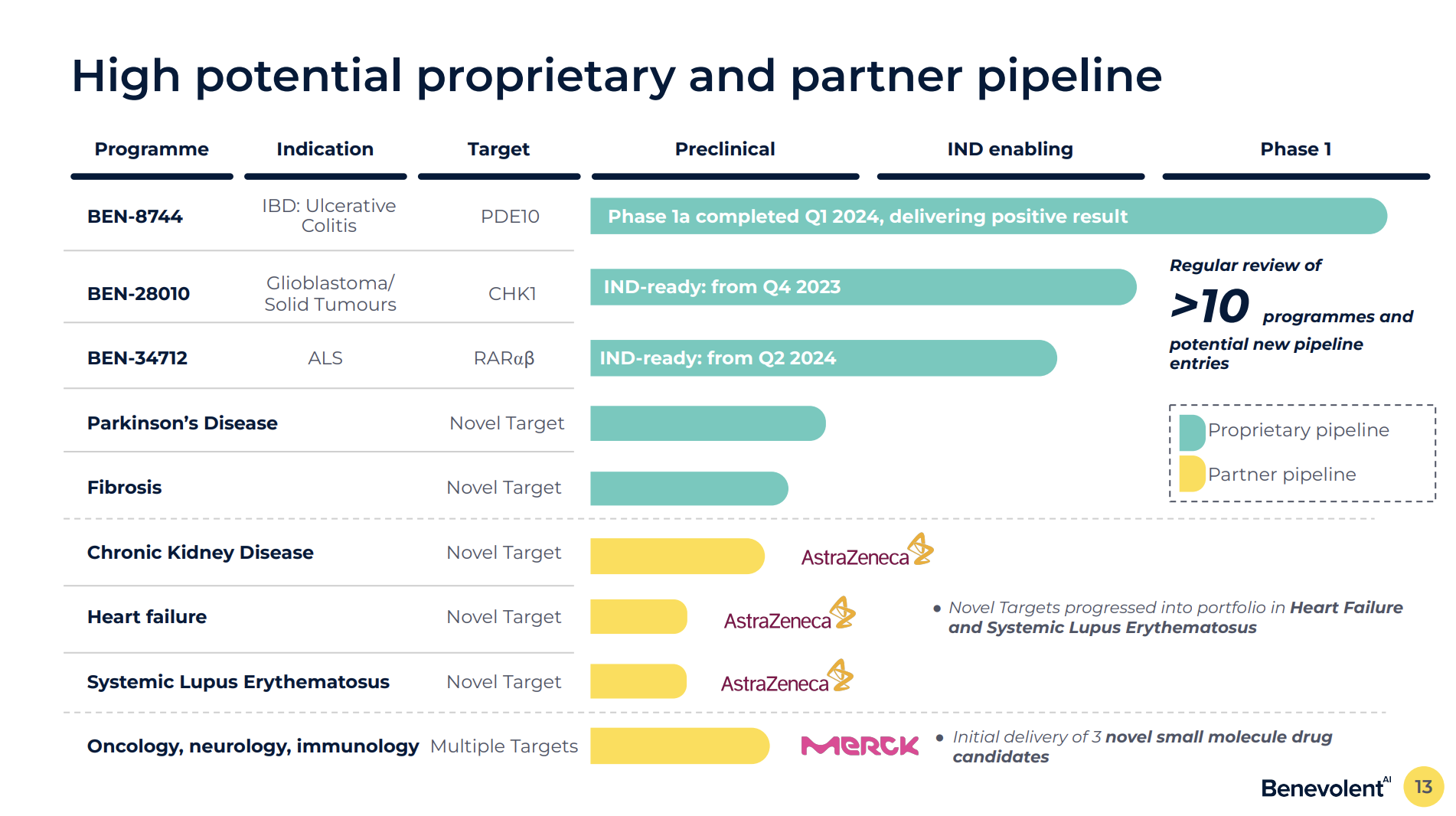
The platform’s credibility rests on collaborations such as a multi‑target AstraZeneca alliance, where several AI‑identified targets have been accepted into development portfolios, and a Merck partnership worth up to $594 million spanning oncology, neurology and immunology. BenevolentAI’s early role in identifying baricitinib as a COVID‑19 treatment, later validated in trials and recognised by the 2020 Scrip Innovation Award, further anchors its evidence base.
From a buyer’s perspective, BenevolentAI is best suited to teams looking to reshape upstream portfolio strategy: which mechanisms to go after, how to rank targets and indications, and where to find repurposing opportunities. It does not aim to replace clinical judgment or downstream development processes, and its listing stresses that AI‑generated hypotheses must be validated scientifically and clinically, with attention to data quality, regulatory review and integration into existing R&D systems.
Atomwise: structure‑based virtual screening at scale
Atomwise (now trading as Numerion Labs) focuses squarely on small‑molecule discovery, using its AtomNet deep learning model to replace or augment traditional high‑throughput screening. The platform evaluates 3D protein–ligand interactions across very large virtual libraries to prioritise hits, helping teams move from millions or billions of candidates down to tens or hundreds of compounds worth synthesising and testing. Atomwise is most persuasive when the task is to turn vast chemical space into a manageable set of testable compounds. The figure below shows why: across crystal structures, cryo-EM and homology models, AtomNet maintained similar hit rates, suggesting the platform can still deliver useful results even when structural data are imperfect.
Figure 2: Atomwise hit rates across crystal structures, cryo-EM and homology models [3]
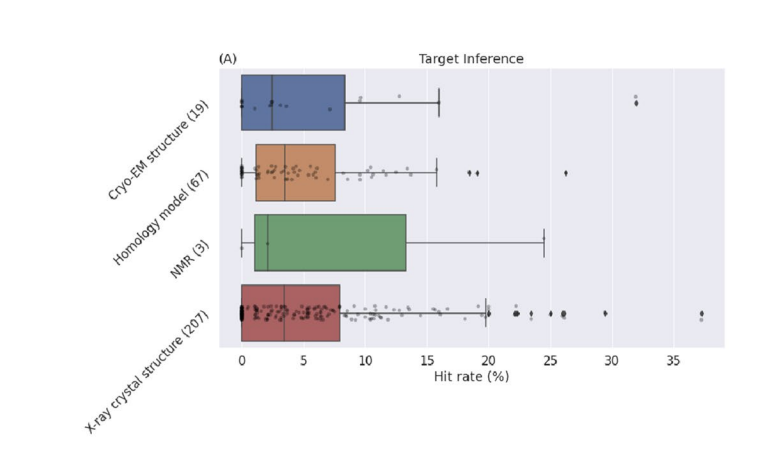
A key evidence pillar is a prospective 318‑target study across more than 250 labs, where AtomNet‑guided campaigns reported project‑level success around 73–75% and hit rates of 5–7.6%, including for targets with little or no prior ligand data, suggesting it can handle “dark” proteins as well as familiar ones. Large collaborations, such as a Sanofi deal with up to approximately $1.2 billion in milestones and further alliances with Eli Lilly, Bayer, Hansoh and others, signal repeated commercial validation.
For buyers, Atomwise is most compelling if you have structurally tractable targets and want to compress design–make–test cycles and reduce plate‑based HTS load, while still keeping medicinal chemistry and assay design in house. The listing emphasises that success depends on high‑quality structural and assay data, robust integration into cheminformatics and ELN environments, and disciplined governance so teams understand that AtomNet scores guide, but do not replace, scientific judgment.
Insilico: integrated generative AI and autonomous lab
Insilico positions itself as a clinical‑stage generative AI drug discovery company with a tightly integrated stack that spans target discovery (PandaOmics), de novo molecule design (Chemistry42), clinical outcome prediction (inClinico) and a fully automated Life Star robotics lab. This closed‑loop design–make–test system is intended to shorten timelines from biological hypothesis to clinical‑stage candidates by continuously feeding experimental data back into AI models. That closed-loop model is visible in Insilico’s TNIK program, which moved from AI-driven discovery into clinical-stage development. The timeline below shows how Insilico progressed TNIK from target discovery and chemistry AI through preclinical work and into Phase 2a.
Figure 3: Insilico TNIK Program Timeline [4]
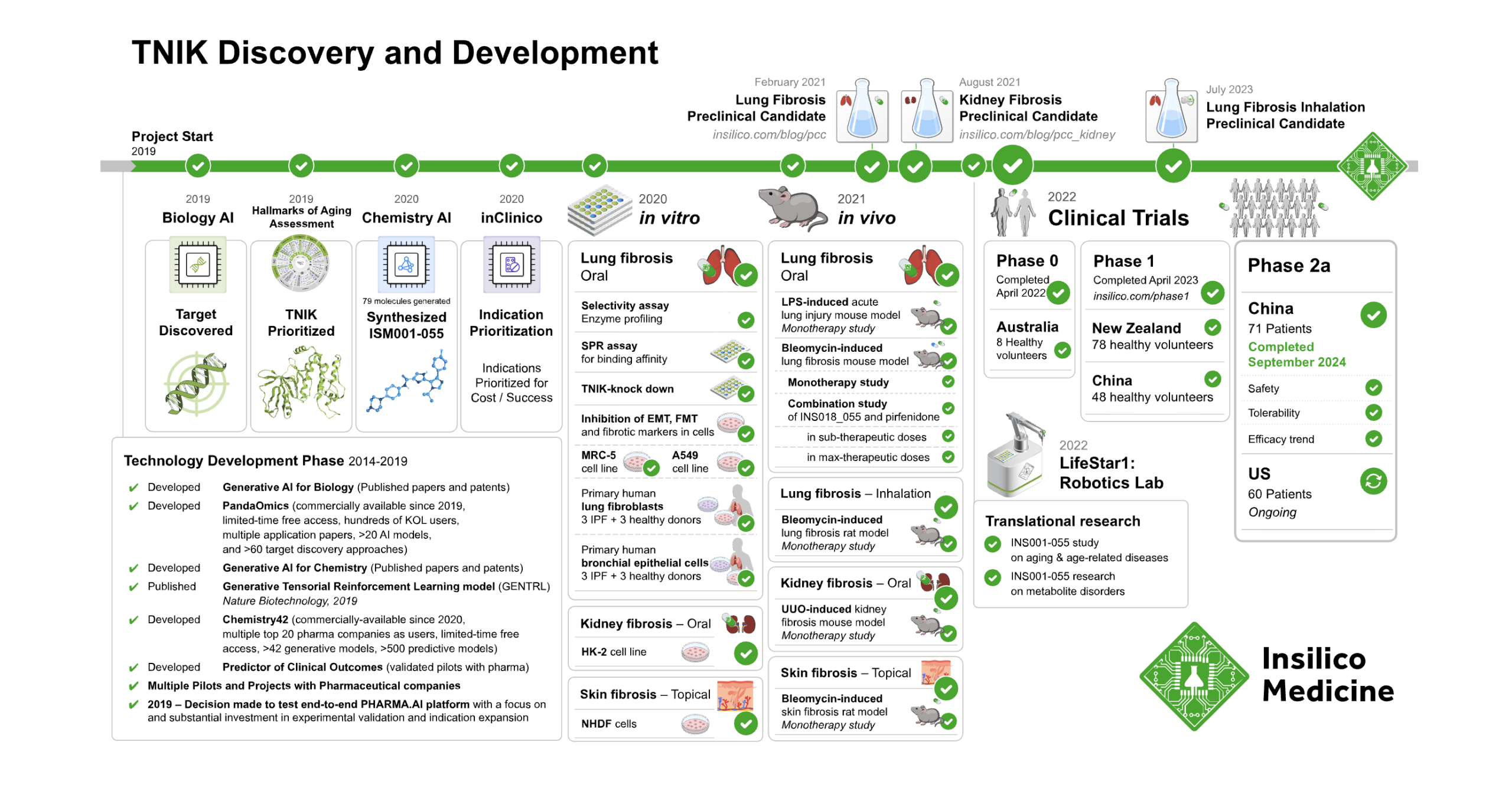
Unlike platforms that stop at discovery, Insilico’s directory entry highlights multiple AI‑designed molecules in human trials, including ISM001‑055 (Rentosertib) for idiopathic pulmonary fibrosis, which reached Phase IIa with encouraging safety and dose‑response signals, and additional programmes in inflammatory bowel disease and oncology. A pipeline of more than 30 programs, at least 10 IND clearances and collaborations with partners such as Servier and Eli Lilly support the claim that the platform is delivering pipeline‑grade assets, not just in‑silico concepts.
Insilico is therefore best aligned with buyers looking for an end‑to‑end AI partner or platform, particularly large pharma and capitalised biotechs seeking to run multiple AI‑enabled programmes in parallel. At the same time, the listing underscores that platform‑level GxP validation and regulated clinical‑decision support status are not established, so organisations must treat Pharma.AI as a powerful but unregulated tool that needs to sit inside their own governance and validation frameworks.
Which AI tool in drug discovery makes sense for you?
For large pharma companies with established discovery engines and growing in-house AI teams, the main question is where an external platform should plug into the existing stack. BenevolentAI is a strong fit when the priority is upstream biology and portfolio-level decision-making: identifying mechanisms, prioritising indications and surfacing repurposing opportunities from literature, omics and clinical data.
Atomwise is a better fit when the biology is reasonably clear but the bottleneck is screening capacity. It suits teams with structural biology, cheminformatics and assay capabilities in place, where the goal is to improve hit quality and reduce time and cost in virtual screening and hit identification rather than outsource the full discovery workflow.
Insilico is most relevant where the buyer is open to deeper program-level partnership, from target discovery through to clinical candidates. It is attractive for large pharma and well-capitalised biotechs that want an integrated AI partner with clinical-stage assets, but it also requires clear governance around data, IP and how AI-generated candidates move into the internal development organisation.
For mid-size and emerging biotechs, the same platforms map to different constraints. BenevolentAI can act as an external biology engine, Atomwise can provide focused virtual screening support when internal infrastructure is limited, and Insilico can add an end-to-end discovery partnership when capital and shared-program appetite are in place. The right choice depends less on the branding of the platform and more on where your team is strongest, where the bottleneck sits and how much of the workflow you want to retain in house.
None of these platforms is a fit for teams looking to outsource scientific judgment, skip internal validation, or replace an established discovery workflow entirely.
Want to compare more AI drug discovery tools?
Explore our Drug Discovery category for more platform comparisons, buyer guides and focused analysis.

Author: Stephen
Founder of HealthyData.Science · 20+ years in life sciences compliance & software validation · MSc in Data Science & Artificial Intelligence.
- This drug discovery directory provides buyer-grade due diligence by synthesizing technical evidence on AI tools for target identification and lead optimization. The platform evaluates solutions based on scientific validity and reproducibility to help R&D leaders de-risk candidates before preclinical and clinical investment. HealthyData.Science. (2026). Drug Discovery: A Curated Directory of AI Solutions and Evidence-Led Vendor Profiles.[↩]
- BenevolentAI’s pipeline includes the PDE10 inhibitor BEN-8744 for ulcerative colitis, the CHK1 inhibitor BEN-28010 for glioblastoma, and the RARαβ agonist BEN-34712 for ALS. The company’s portfolio also features novel targets in heart failure and lupus through validated collaborations with AstraZeneca and Merck. BenevolentAI. (2024). Investor Presentation: August 2024 Update on Clinical Pipeline and Strategic Collaborations.[↩]
- Atomwise demonstrated consistent hit rates across varied structural inputs, including X-ray, cryo-EM, and homology models. The company’s AtomNet platform achieved average hit rates of 5.6% for crystal structures and 5.1% for homology models, confirming its robustness in identifying active molecules even with lower-precision target data. Stecula, A., et al. (2024). Performance of AtomNet across varied structural inputs: Hit rates in X-ray, cryo-EM, and homology models. Scientific Reports.[↩]
- Insilico Medicine’s TNIK program demonstrates an integrated AI-driven workflow from target discovery to clinical validation. Using the Pharma.AI platform and robotics, the company advanced ISM001-055 from inception to Phase 2a trials for idiopathic pulmonary fibrosis, providing clinical-stage evidence for its generative AI drug discovery pipeline. Insilico Medicine. (2024). Insilico Medicine announces positive topline results of ISM001-055 for the treatment of idiopathic pulmonary fibrosis (IPF) developed using generative AI.[↩]

